ASAPprime® Regulatory
ASAPprime® Regulatory Interface
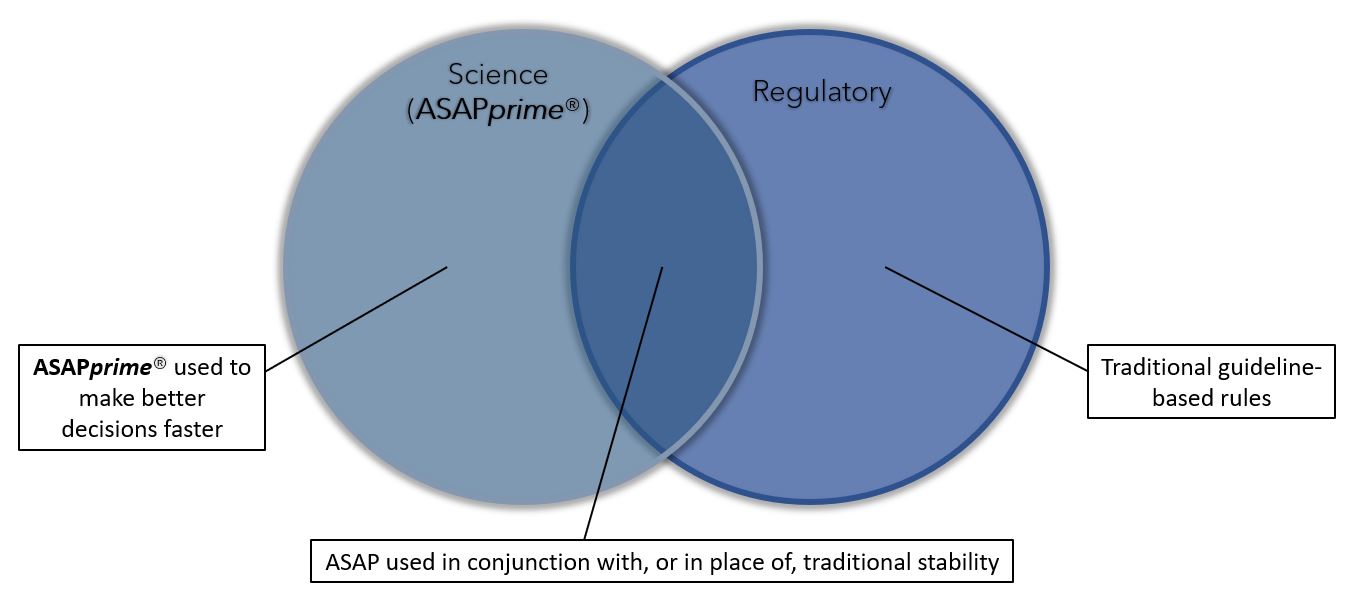
ASAPprime® is used in conjunction with, or as a replacement for, traditional ICH or accelerated stability studies.
There have been over 100 regulatory filings using ASAPprime®. Of these, the majority have been for INDs/IMPDs. However, there have been a number of regulatory submissions filed for new market applications and for post-approval changes.
ASAPprime® has been successfully used in place of traditional stability, in support of use-period assignments for INDs for many country authorities including the USA FDA.
Among the countries that have accepted this are: Belgium, Brazil, Bulgaria, Canada, China, Denmark, Egypt, Finland, France, Georgia, Hungary, India, Ireland, Italy, Lebanon, Netherlands, Poland, Portugal, Russia, Spain, Sweden, Taiwan, Turkey, UK, & Ukraine.
Late stage minor changes have been justified by ASAPprime® as part of NDA filings. ASAPprime® modeling is first validated against the prior ICH data for the product before the change, then ASAPprime® is used to show that the stability does not vary with the change. This allows ASAPprime® to bridge data before and after a process, formulation, or other change.
ASAPprime® has been successfully used to justify post-approval changes of products that would normally require new stability programs (e.g., a reduction in package protection).
As an example, an alternative low strength of a marketed tablet product used ASAPprime® in combination with only 3 months of traditional stability. A 3-year shelf-life was accepted with no questions in the US, UK, France, Italy, Turkey, Egypt, Lebanon, and Kenya.
With generics, ASAPprime® justifies equivalence to the originator.
ASAPprime® can model the impact of excursions, and thereby provide justification for acceptability of products subjected to even harsh conditions for short times.
Regulatory Document Preparation Service
FreeThink offers pharmaceutical customers a service which includes guidance on how to leverage ASAPprime® in regulatory filings. FreeThink can support the preparation of Common Technical Document (CTD) IND/IMPD, NDA, and MAA sections related to stability and shelf life justification, including, but not limited to drug substance sections under S.7 and drug product sections under P.8.
FreeThink offers help with completing modules in the CTD for investigational (IND/IMPD), commercial, and post-approval change submissions (such as SUPAC) for NDA and ANDA products. This includes the Quality (CMC) sections for the submission captured in ICH M4Q, and sections related to stability data and shelf life justification which generally fall under the Quality sections of the CTD as follows: Drug Substance (3.2.S.7 Stability) and Drug Product (3.2.P.8 Stability).
FreeThink also supports any further elaboration needed on stability and ASAPprime® stability projections of the drug product development sections of the CTD:
Drug Product
- 3.2.P.2.1 Components of the drug product (drug substance and excipients)
- 3.2.P.2.2 Formulation Development
- 3.2.P.2.4 Container Closure Systems
For new drug applications (NDA and ANDA) and for post-approval changes where the use of ASAPprime® might support future product changes, US/FDA Comparability Protocols (CP) or ICH (Q12), Post Approval Change Management Protocol (PACMP) may be used to lay out a plan to use ASAPprime®. These documents are filed in the Regional Section of the Quality Module: 3.2.R. Regional Information.
Resources
For more information, see the following resources:
E. Debie “Risk-Based Predictive Stability: Feedback from IQ Working Group Regulatory Sub-Team”
Presented at Science of Stability Conference, Amsterdam, the Netherlands, October 2019
F. Qiu, et al. “Accelerated Predictive Stability: Fundamentals and Pharmaceutical Industry Practices”
This book discusses many aspects of ASAP, with case studies.
© 2018



